Pfizer is asking U.S. regulators to authorize its updated COVID-19 vaccine for children under age 5 — not as a booster but part of their initial shots.
The youngest tots already are supposed to get three extra-small doses of the original vaccine as their primary series. Pfizer and its partner BioNTech said Monday, Dec. 5, 2022 that if the Food and Drug Administration agrees, the updated vaccine would be used for the third shot. The FDA already has cleared COVID-19 vaccines tweaked to better target omicron as boosters for everyone 5 and older.
Children ages 6 months through 4 years already are supposed to get three extra-small doses of the original Pfizer COVID-19 vaccine — each a tenth of the amount adults receive — as their primary series. If the Food and Drug Administration agrees, a dose of Pfizer's bivalent omicron-targeting vaccine would be substituted for their third shot.
Pfizer and its partner BioNTech said Monday that may help prevent severe illness and hospitalization from COVID-19 in little kids, at a time when children’s hospitals already are packed with youngsters hit by other respiratory illnesses.: Just 2% of tots under 2 and about 4% of 2- to 4-year-olds have gotten their primary doses so far, according to the Centers for Disease Control and Prevention.Those combination shots contain half the original vaccine and half tweaked to match the BA.4 and BA.
Copyright 2022 The Associated Press. All rights reserved. This material may not be published, broadcast, rewritten or redistributed without permission.
México Últimas Noticias, México Titulares
Similar News:También puedes leer noticias similares a ésta que hemos recopilado de otras fuentes de noticias.
 Pfizer/BioNTech seek FDA authorization of updated Covid-19 vaccine for children under 5 | CNNPfizer and BioNTech have submitted an application to the US Food and Drug Administration for their updated Covid-19 vaccine to be used as the third shot in the three-dose primary vaccine series for children ages 6 months through 4 years.
Pfizer/BioNTech seek FDA authorization of updated Covid-19 vaccine for children under 5 | CNNPfizer and BioNTech have submitted an application to the US Food and Drug Administration for their updated Covid-19 vaccine to be used as the third shot in the three-dose primary vaccine series for children ages 6 months through 4 years.
Leer más »
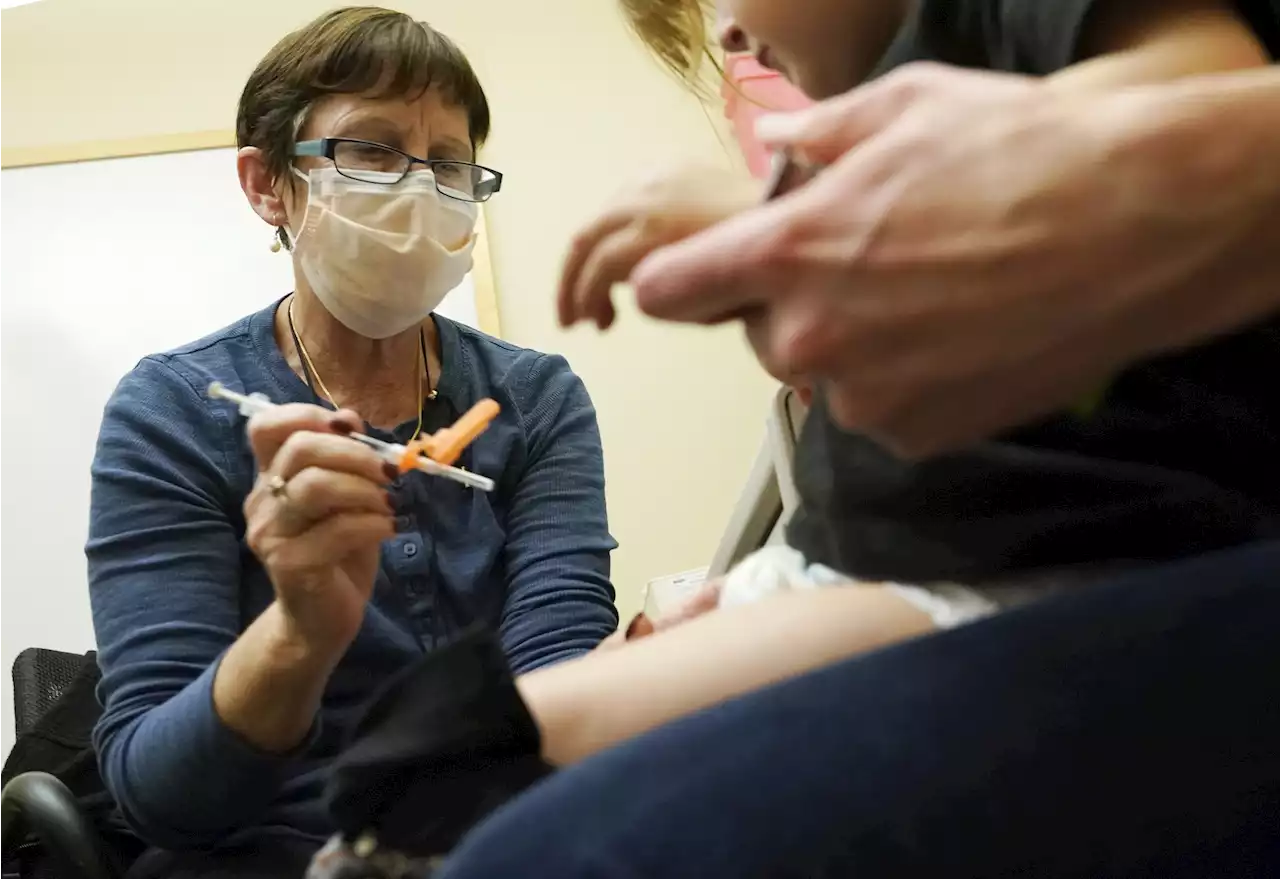 Pfizer asks FDA to clear updated COVID shot for kids under 5Pfizer is asking U.S. regulators to authorize its updated COVID-19 vaccine for children under age 5 — not as a booster but part of their initial shots. Children ages 6 months through 4 years already are supposed to get three extra-small doses of the original Pfizer COVID-19 vaccine — each a tenth of the amount adults receive — as their primary series.
Pfizer asks FDA to clear updated COVID shot for kids under 5Pfizer is asking U.S. regulators to authorize its updated COVID-19 vaccine for children under age 5 — not as a booster but part of their initial shots. Children ages 6 months through 4 years already are supposed to get three extra-small doses of the original Pfizer COVID-19 vaccine — each a tenth of the amount adults receive — as their primary series.
Leer más »
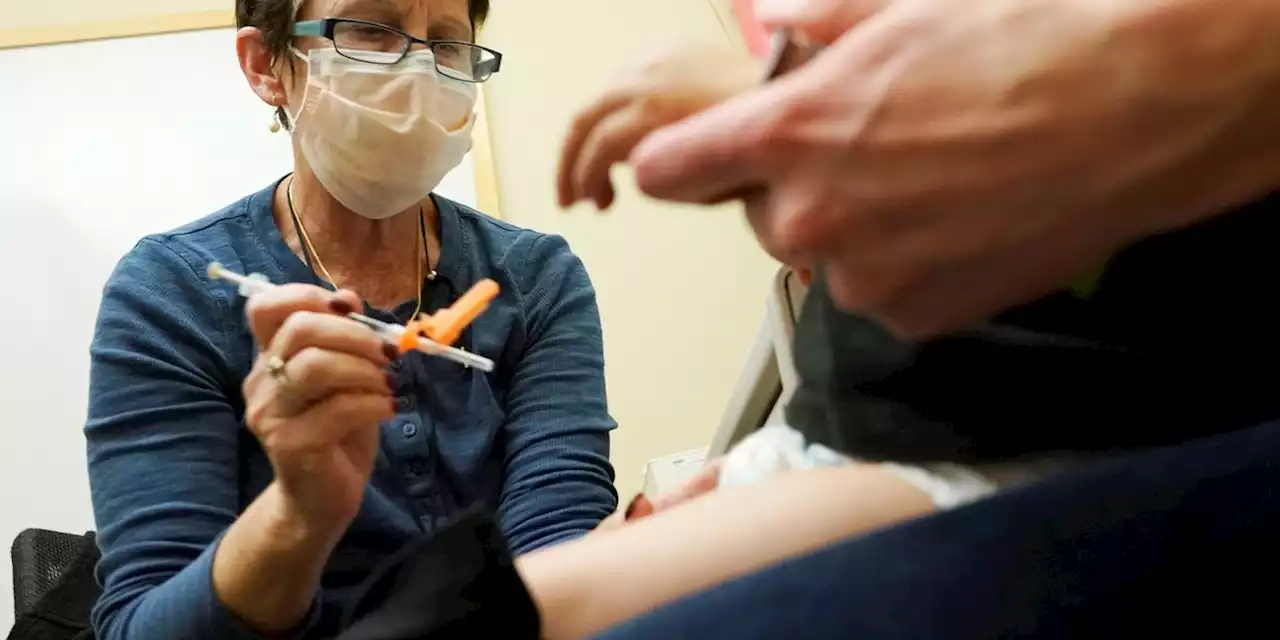 Pfizer asks FDA to clear updated COVID shot for kids under 5Pfizer and its partner BioNTech said Monday that may help prevent severe illness and hospitalization from COVID-19 in little kids, at a time when children’s hospitals already are packed with youngsters hit by other respiratory illnesses.
Pfizer asks FDA to clear updated COVID shot for kids under 5Pfizer and its partner BioNTech said Monday that may help prevent severe illness and hospitalization from COVID-19 in little kids, at a time when children’s hospitals already are packed with youngsters hit by other respiratory illnesses.
Leer más »
 Pfizer asks FDA to clear updated COVID shot for kids under 5Pfizer is asking U.S. regulators to authorize its updated COVID-19 vaccine for children under age 5.
Pfizer asks FDA to clear updated COVID shot for kids under 5Pfizer is asking U.S. regulators to authorize its updated COVID-19 vaccine for children under age 5.
Leer más »
 Pfizer, BioNTech ask FDA to authorize bivalent COVID vaccine in young childrenPfizer Inc. undefined and BioNTech SE undefined said Monday that they asked the Food and Drug Administration to authorize their updated COVID-19 vaccine as...
Pfizer, BioNTech ask FDA to authorize bivalent COVID vaccine in young childrenPfizer Inc. undefined and BioNTech SE undefined said Monday that they asked the Food and Drug Administration to authorize their updated COVID-19 vaccine as...
Leer más »
 Pfizer asks FDA to clear updated COVID shot for kids under 5Pfizer and its partner BioNTech said Monday that may help prevent severe illness and hospitalization from COVID-19 in little kids, at a time when children’s hospitals already are packed with youngsters hit by other respiratory illnesses.
Pfizer asks FDA to clear updated COVID shot for kids under 5Pfizer and its partner BioNTech said Monday that may help prevent severe illness and hospitalization from COVID-19 in little kids, at a time when children’s hospitals already are packed with youngsters hit by other respiratory illnesses.
Leer más »
